Complicated, hard-to-heal wounds are a growing medical problem and there are currently only two drugs approved with proven efficacy. In a new study on humans, researchers at Uppsala University show that treatment with a specific type of modified lactic acid bacteria works well and has a positive effect on the healing of wounds.
In several controlled preclinical models, the research team behind the new study has previously demonstrated accelerated wound healing after topical treatment (treatment on the skin) using lactic acid bacteria, or Limosilactobacillus reuteri, genetically modified to produce the chemokine CXCL12 (ILP100-Topical).
The researchers can now show data from the first clinical study on humans, in which the main objective was to establish safety and tolerability. Other objectives were to see clinical and biological effects on wound healing using traditionally accepted methods, as well as more exploratory and traceable measurements. 36 healthy volunteers were included in the study with a total of 240 induced wounds studied. The study’s design and methodology are described in more detail below.
The results show that treatment using ILP100-Topical was safe and well tolerated among all individuals and doses, and neither ILP100 nor CXCL12 could be detected in locations beyond the wounds. A significantly higher proportion of healed wounds (p=0.020) was seen on day 32 using multi-dose ILP100-Topical compared to saline and placebo (76% (73/96) and 59% (57/96) healed wounds respectively) when the results from the multi-dose-treated wounds were pooled.
In addition, the time to first recorded healing was reduced by an average of 6 days, and by 10 days at the highest dose. The mechanism of action of ILP100-Topical was also confirmed when the treatment resulted in increased CXCL12-positive cells in the wounds, as well as increased blood flow around the wounds during the healing phase.
“Our study shows that bacteria modified to produce and deliver human protein for local effects can be used as drugs to accelerate the healing of wounds. This is the first time this has been shown in controlled human studies, and it can be expected that the effect is greater in patients with diseases that negatively affect wound healing,” explains Mia Phillipson, Professor at the Department of Medical Cell Biology at Uppsala University.
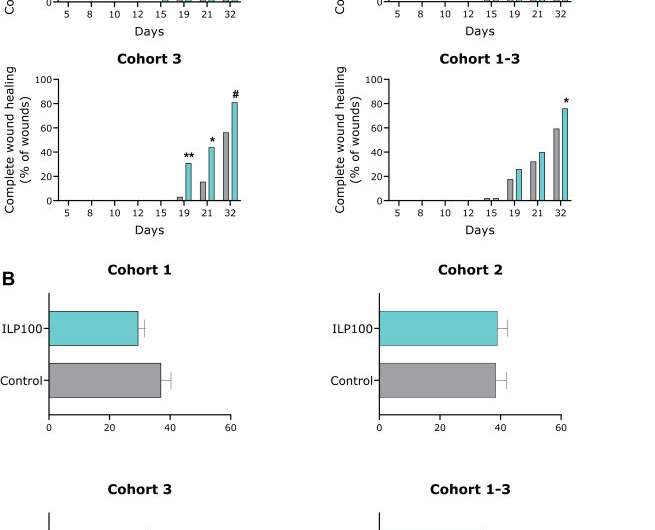
The favorable safety profile and the beneficial effects on wound healing observed here support further clinical development of ILP100-Topical for the treatment of complex and hard-to-heal wounds in patients, which is already under way.
Many immune-active proteins are inherently unstable and degrade quickly, so supplying them from lactic acid bacteria to the exact site of action is one way to develop them as drugs.
“The potential is really endless when you consider how important a role proteins play in various processes in the body, and how many diseases we currently do not have good enough treatments for. We have already produced another drug candidate to cure and reduce inflammation in the gut of cancer patients—ILP100-Oral—and in the future we will start a research project with another chemokine for the treatment of lung diseases,” concludes Phillipson.
SITU-SAFE is an adaptive, randomized, double-blind, placebo-controlled, phase 1 study (EudraCT 2019-000680-24) consisting of a single ascending dose (SAD) part for each participant and a multiple ascending dose (MAD) part for each participant. Both parts of the study each consisted of three dose cohorts.
A total of 240 wounds were induced on the upper arms of 36 healthy volunteers, including 4 wounds (2/arm) in 12 participants in the SAD arm, and 8 wounds (4/arm) in 24 participants in the MAD arm. The wounds were randomized for treatment using a placebo, saline or ILP100-Topical. The study was conducted at the Phase 1 unit at Uppsala University Hospital in Uppsala, Sweden, and sponsored by Ilya Pharma.
The research is published in the journal eClinicalMedicine.
More information:
Emelie Öhnstedt et al, Engineered bacteria to accelerate wound healing: an adaptive, randomised, double-blind, placebo-controlled, first-in-human phase 1 trial, eClinicalMedicine (2023). DOI: 10.1016/j.eclinm.2023.102014
Journal information:
EClinicalMedicine
Source: Read Full Article