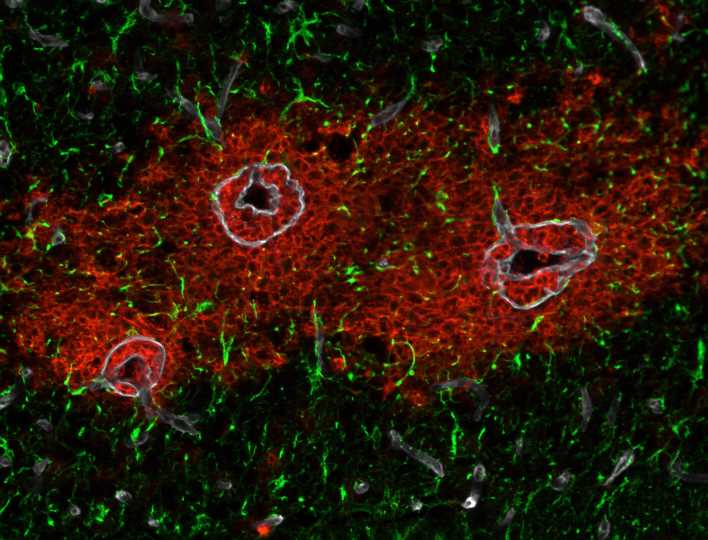
Researchers have identified novel gelatinase substrates involved in astroglial barrier function. In neuroinflammation, immune cells such as leukocytes cross the blood-brain barrier. One key to this ability is the gelatinases matrix metalloproteinase MMP-2 and MMP-9. Until now, the substrates of the enzymes involved in the process were unknown.
Now, using a sensitive mass spectrometry–based secretome approach, researchers at the University of Münster and Bonn University Hospital (UKB) have succeeded in identifying hundreds of such molecules that are cleaved from the cell surface of astrocytes. In doing so, they have generated a unique database of MMP-2/-9 substrates specific to the formation and maintenance of the barrier as well as communication between astrocytes and neurons. The results have been published in the journal Science Advances.
The endothelial cells on the inner wall of cerebral blood vessels form a protective barrier to the brain via tightly linked junctions. However, without the underlying astrocytes, a form of glial cells, a fully functional blood-brain barrier (BBB) cannot form.
In neuroinflammation, the endothelial and astroglial layers are molecularly and functionally two distinct barriers to invading white blood cells, known as leukocytes. However, studies in multiple sclerosis (MS) show that disease symptoms do not develop until immune cells have also penetrated the astroglial layer.
“This underscores their important contribution to the functional integrity of the BBB as well as their independence from the endothelial barrier,” says Prof. Lydia Sorokin, director of the Institute of Physiological Chemistry and Pathobiochemistry at the University of Münster. “But in contrast to leukocyte penetration of the endothelial barrier, there has been little knowledge of subsequent processes at the astroglial layer.”
It is known that the gelatinases, MMP-2 and MMP-9 regulate the invasion of leukocytes into the brain during neuroinflammation. Activity of these two protein-cleaving enzymes is thus an early marker of invasion of the brain parenchyma by these immune cells—to date, the only specific marker of ongoing neuroinflammation.
“Evidence suggests that MMP-2 and MMP-9 have both positive and negative effects on the BBB. Therefore, deciphering their substrate specificity at the brain parenchymal boundary will contribute to the understanding of molecular processes essential for astroglial barrier function,” said Sorokin.
Identifying the enzyme cleavage sites is a challenge. The research team relied on recent advances in, among other things, mass spectrometry (MS) to analyze the secretome—a method that can comprehensively detect proteins secreted by cells. In this study, they further developed this method to identify proteolytic cleavages of cell membrane-associated proteins.
“Our approach detects extracellularly released protein fragments independently of biochemical enrichments and is therefore particularly sensitive,” said Prof. Felix Meissner, director of the UKB’s Institute of Innate Immunity. Using a tailored secretome MS approach, the team identified two major classes of compounds released by MMP-2/MMP-9 from the astrocyte cell surface. Validation of these novel substrates of neuroinflammation was performed in the mouse model of multiple sclerosis and in human MS samples.
Overall, the combination of the secretome MS approach with knowledge of the astroglial barrier provides a unique database of previously unknown gelatinase substrates that likely contribute to the barrier function of the astroglial boundary. In addition, evidence suggests that MMP-2/MMP-9 activity may also influence communication between astrocytes and neurons.
“Our approach to identify proteolytic processes that control astroglial barrier function works and provides opportunities for future research to understand the molecular nature of the astroglial barrier and its contribution to the BBB,” said Meissner.
More information:
Miriam Burmeister et al, Secretomics reveals gelatinase substrates at the blood-brain barrier that are implicated in astroglial barrier function, Science Advances (2023). DOI: 10.1126/sciadv.adg0686
Journal information:
Science Advances
Source: Read Full Article