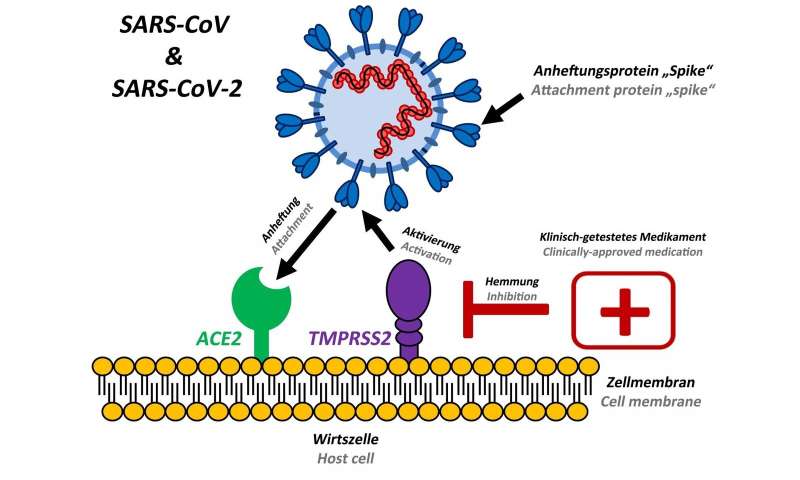
Several coronaviruses circulate worldwide and constantly infect humans, which normally causes only mild respiratory disease. Currently, however, we are witnessing a worldwide spread of a new coronavirus with more than 90,000 confirmed cases and over 3,000 deaths. The new virus has been named SARS coronavirus-2 and has been transmitted from animals to humans. It causes a respiratory disease called COVID-19 that may take a severe course. The SARS coronavirus-2 has been spreading since December 2019 and is closely related to the SARS coronavirus that caused the SARS pandemic in 2002/2003. No vaccines or drugs are currently available to combat these viruses.
Stopping virus spread
A team of scientists led by infection biologists from the German Primate Centre and including researchers from Charité, the University of Veterinary Medicine Hannover Foundation, the BG-Unfallklinik Murnau, the LMU Munich, the Robert Koch Institute and the German Center for Infection Research, wanted to find out how the new coronavirus SARS-CoV-2 enters host cells and how this process can be blocked. The researchers identified a cellular protein that is important for the entry of SARS-CoV-2 into lung cells. “Our results show that SARS-CoV-2 requires the protease TMPRSS2, which is present in the human body, to enter cells,” says Stefan Pöhlmann, head of the Infection Biology Unit at the German Primate Center. “This protease is a potential target for therapeutic intervention.”
Promising drug
Since it is known that the drug camostat mesilate inhibits the protease TMPRSS2, the researchers have investigated whether it can also prevent infection with SARS-CoV-2. “We have tested SARS-CoV-2 isolated from a patient and found that camostat mesilate blocks entry of the virus into lung cells,” says Markus Hoffmann, the lead author of the study. Camostat mesilate is a drug approved in Japan for use in pancreatic inflammation. “Our results suggest that camostat mesilate might also protect against COVID-19,” says Markus Hoffmann. “This should be investigated in clinical trials.”
Source: Read Full Article
